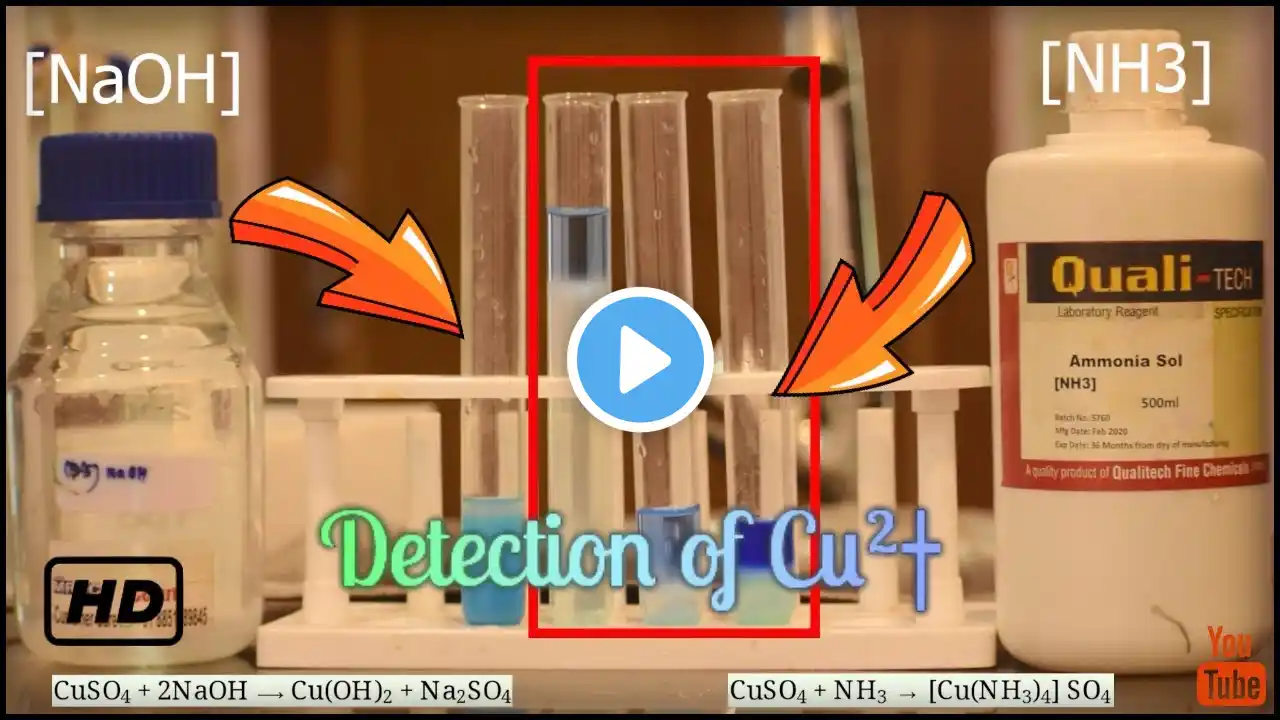
Quick and easy detection of nickel metal ions in coins
The video shows a few drops of dimethylglyoxime, C4H6(NOH)2, being added to a dilute solution of nickel(II) sulfate with a few drops of ammonia solution. Immediately, an insolube red solid called nickel dimethylglyoxime, Ni(C4H8N2O2)2, precipitates out of solution. The balanced ionic chemical equation for this reaction: Ni2+(aq) + 2C4H8N2O2(aq)→ Ni(C4H7N2O2)2(s)↓+ 2H+(aq) The reaction involves two dimethylglyoxime molecules acting as chelating agents to form the nickel dymethylglyoxime complex. This reaction is very sensitive and can be used as a confirmation test for the presence of nickel(II) cations even in very low concentrations. This reaction can be easily applied to metal objects that regularly come in contact with the skin, for example coins, jewellery, stainless steel cutlery, spectacle frames, watch straps, etc. for the benefit of people that suffer from a kind of skin sensitivity or dermatitis called Nickel itch. This contact allergy flares up when sweating, the acids present dissolve a little of the nickel and can accentuate conditions such as skin erythema, eczema, etc. It is also possible to identify meteorites because of their high nickel content compared to ore samples. Even though commercial products exist to test metals for the presence of nickel a much simpler and cheap version can be made at home with household ingredients. First, obtain some cotton buds and dip them in a solution of 1 % dimethylglyoxime (DMG) in ethanol (vodka will work just as well) for 10 minutes. NOTE: DMG is flammable, irritant and the only chemical that is difficult to obtain but very small amounts (1 g approx.) is needed to make a large stock of prepared buds. Once they have been soaking for at least 10 minutes, remove them and leave them to air dry. They are now ready to be used or can be stored in an air tight container labelled appropriately. To test for the presence of nickel in the desired metal object add a few drops of dilute ammonia solution (household ammonia will work depending on concentration, higher concentrations produces faster results) to just wet the tip of the cotton. Clean the article with a moist tissue first and rub the tip on the coin or object for about a minute and observe if any pink-red colour appears. The intensity of the colour will depend on how much nickel is present or if it is freely available on the surface of the object (dark pink-red if made completely out of nickel) or light pink if alloyed with other metals. Interestingly, even when bound tightly in an alloy, the test will detect at least some of the nickel cations present. Some alloys more than others depending on composition or quality of production. For an excellent infographic on the composition of UK coins and to use as a comparison please see Compound Interest’s metals in UK coins: https://www.compoundchem.com/2014/03/... For an in depth analysis a control sample colour to compare with could be obtained by using a standard nickel(II) sulfate solution. A standard solution followed by colorimetry analysis of the test samples could be an option to determine nickel concentration in a more quantitative way. REFERENCES: https://beta-static.fishersci.com/con... http://chemlab.truman.edu/files/2015/...