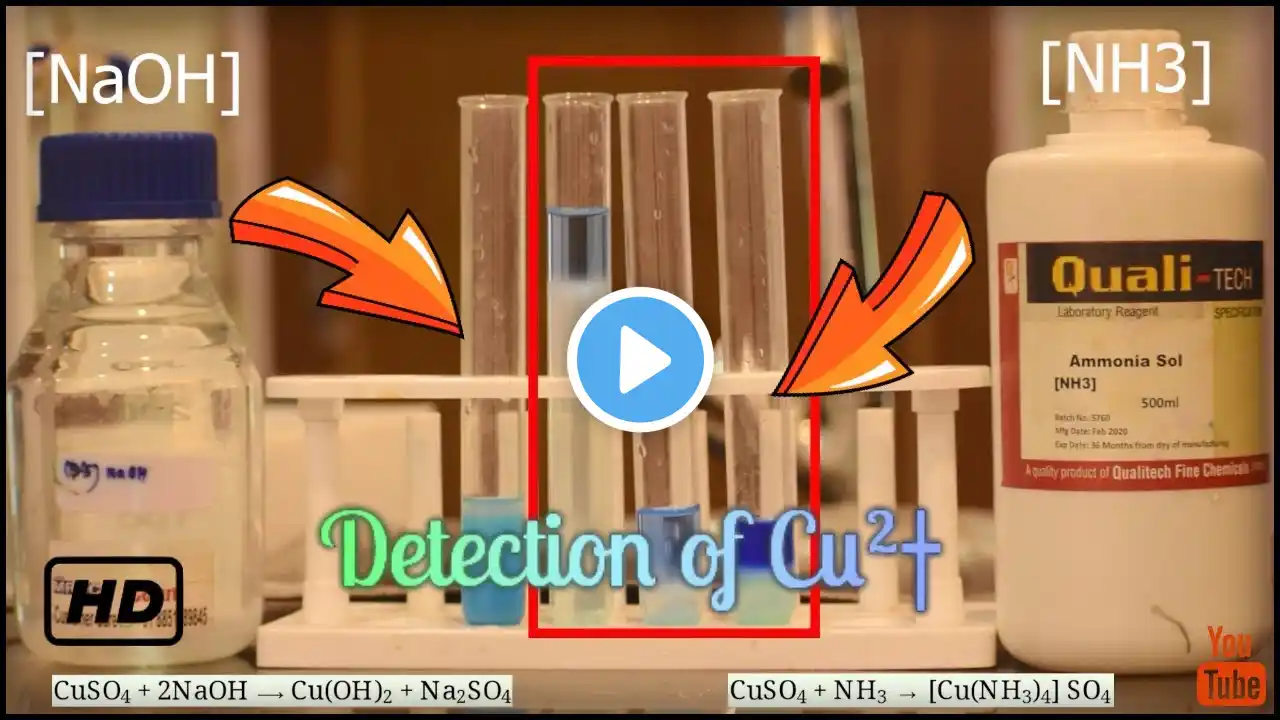
#Detection of Cu(II) || #copper(II)ion detection ||#analytical chemistry || #Bluevitrion
Copper (disambiguation). Copper is a chemical element with the symbol Cu (from Latin: cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orange color. Copper is used as a conductor of heat and electricity, as a building material, and as a constituent of various metal alloys, such as sterling silver used in jewelry, cupronickel used to make marine hardware and coins, and constantan used in strain gauges and thermocouples for temperature measurement. Copper, 29Cu Native copper (~4 cm in size) Copper Appearance red-orange metallic luster Standard atomic weight Ar, std(Cu) 63.546(3)[1] Copper in the periodic table Hydrogen Helium Lithium Beryllium Boron Carbon Nitrogen Oxygen Fluorine Neon Sodium Magnesium Aluminium Silicon Phosphorus Sulfur Chlorine Argon Potassium Calcium Scandium Titanium Vanadium Chromium Manganese Iron Cobalt Nickel Copper Zinc Gallium Germanium Arsenic Selenium Bromine Krypton Rubidium Strontium Yttrium Zirconium Niobium Molybdenum Technetium Ruthenium Rhodium Palladium Silver Cadmium Indium Tin Antimony Tellurium Iodine Xenon Caesium Barium Lanthanum Cerium Praseodymium Neodymium Promethium Samarium Europium Gadolinium Terbium Dysprosium Holmium Erbium Thulium Ytterbium Lutetium Hafnium Tantalum Tungsten Rhenium Osmium Iridium Platinum Gold Mercury (element) Thallium Lead Bismuth Polonium Astatine Radon Francium Radium Actinium Thorium Protactinium Uranium Neptunium Plutonium Americium Curium Berkelium Californium Einsteinium Fermium Mendelevium Nobelium Lawrencium Rutherfordium Dubnium Seaborgium Bohrium Hassium Meitnerium Darmstadtium Roentgenium Copernicium Nihonium Flerovium Moscovium Livermorium Tennessine Oganesson – ↑ Cu ↓ Ag nickel ← copper → zinc Atomic number (Z) 29 Group group 11 Period period 4 Block d-block Electron configuration [Ar] 3d10 4s1 Electrons per shell 2, 8, 18, 1 Physical properties Phase at STP solid Melting point 1357.77 K (1084.62 °C, 1984.32 °F) Boiling point 2835 K (2562 °C, 4643 °F) Density (near r.t.) 8.96 g/cm3 when liquid (at m.p.) 8.02 g/cm3 Heat of fusion 13.26 kJ/mol Heat of vaporization 300.4 kJ/mol Molar heat capacity 24.440 J/(mol·K) Vapor pressure P (Pa) 1 10 100 1 k 10 k 100 k at T (K) 1509 1661 1850 2089 2404 2834 Atomic properties Oxidation states −2, 0,[2] +1, +2, +3, +4 (a mildly basic oxide) Electronegativity Pauling scale: 1.90 Ionization energies 1st: 745.5 kJ/mol 2nd: 1957.9 kJ/mol 3rd: 3555 kJ/mol (more) Atomic radius empirical: 128 pm Covalent radius 132±4 pm Van der Waals radius 140 pm Color lines in a spectral range Spectral lines of copper Other properties Natural occurrence primordial Crystal structure face-centered cubic (fcc)Face-centered cubic crystal structure for copper Speed of sound thin rod (annealed) 3810 m/s (at r.t.) Thermal expansion 16.5 µm/(m⋅K) (at 25 °C) Thermal conductivity 401 W/(m⋅K) Electrical resistivity 16.78 nΩ⋅m (at 20 °C) Magnetic ordering diamagnetic[3] Molar magnetic susceptibility −5.46×10−6 cm3/mol[4] Young's modulus 110–128 GPa Shear modulus 48 GPa Bulk modulus 140 GPa Poisson ratio 0.34 Mohs hardness 3.0 Vickers hardness 343–369 MPa Brinell hardness 235–878 MPa CAS Number 7440-50-8 History Naming after Cyprus, principal mining place in Roman era (Cyprium) Discovery Middle East (9000 BC) Symbol "Cu": from Latin cuprum Main isotopes of copper Isotope Abundance Half-life (t1/2) Decay mode Product 63Cu 69.15% stable 64Cu syn 12.70 h ε 64Ni β− 64Zn 65Cu 30.85% stable 67Cu syn 61.83 h β− 67Zn #youtubehashtags #hashtagsforyoutube #getmoreviews
