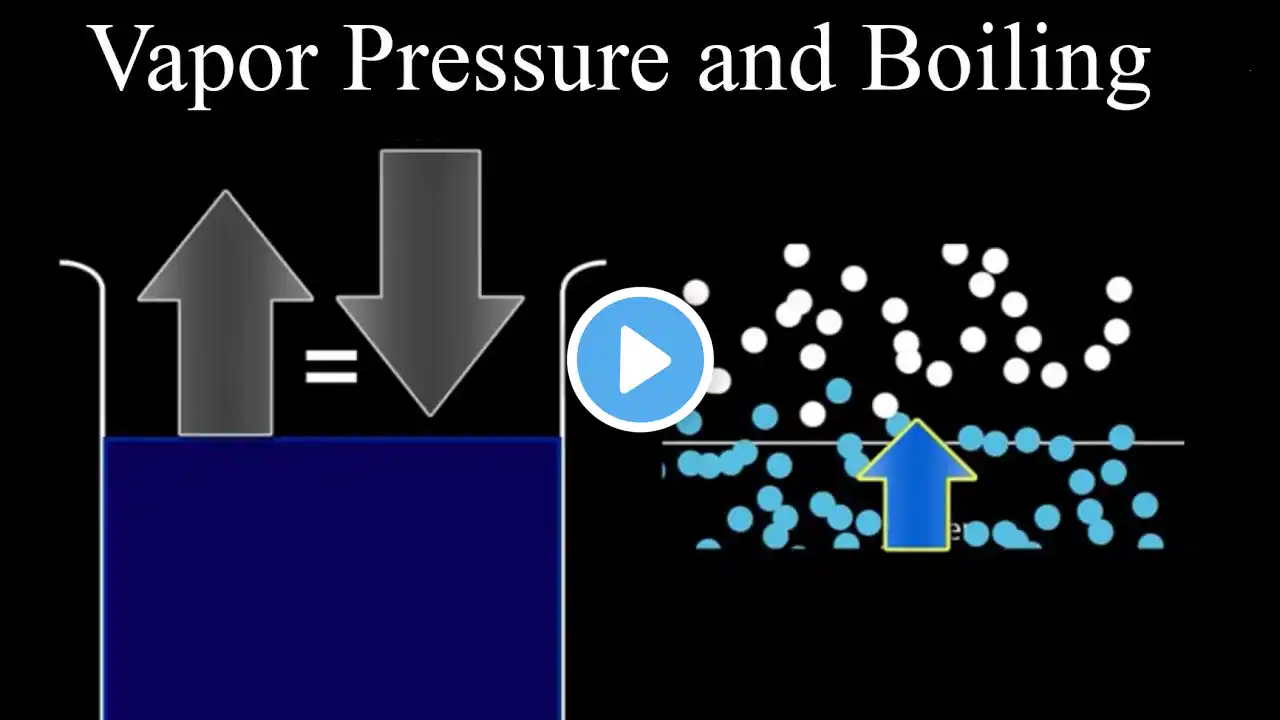
Why Water Boils at Different Temperatures: Pressure, Altitude & Cookers
In this video we explore why water does not always boil at 100 °C and how atmospheric pressure controls the boiling point. You’ll learn the definition of boiling as the moment water’s vapor pressure matches the surrounding air pressure, and see how changes in external pressure shift that balance. The explanation includes: The relationship between vapor pressure, air pressure, and temperature How lower pressure at high altitudes reduces the boiling temperature, affecting cooking times for pasta, beans, and other foods How a pressure cooker increases internal pressure, raising the boiling point and speeding up the preparation of rice, meat, and similar dishes Real‑world demonstrations illustrate these concepts, from mountain‑top cooking experiments to the rapid steam release when a pressure cooker lid is opened. The video also touches on broader applications such as industrial boilers and aircraft coffee makers. By the end, you’ll understand how pressure and boiling point are linked and how this knowledge can be applied in everyday cooking and engineering contexts.