Lecanemab: Breakthrough Alzheimer drug?
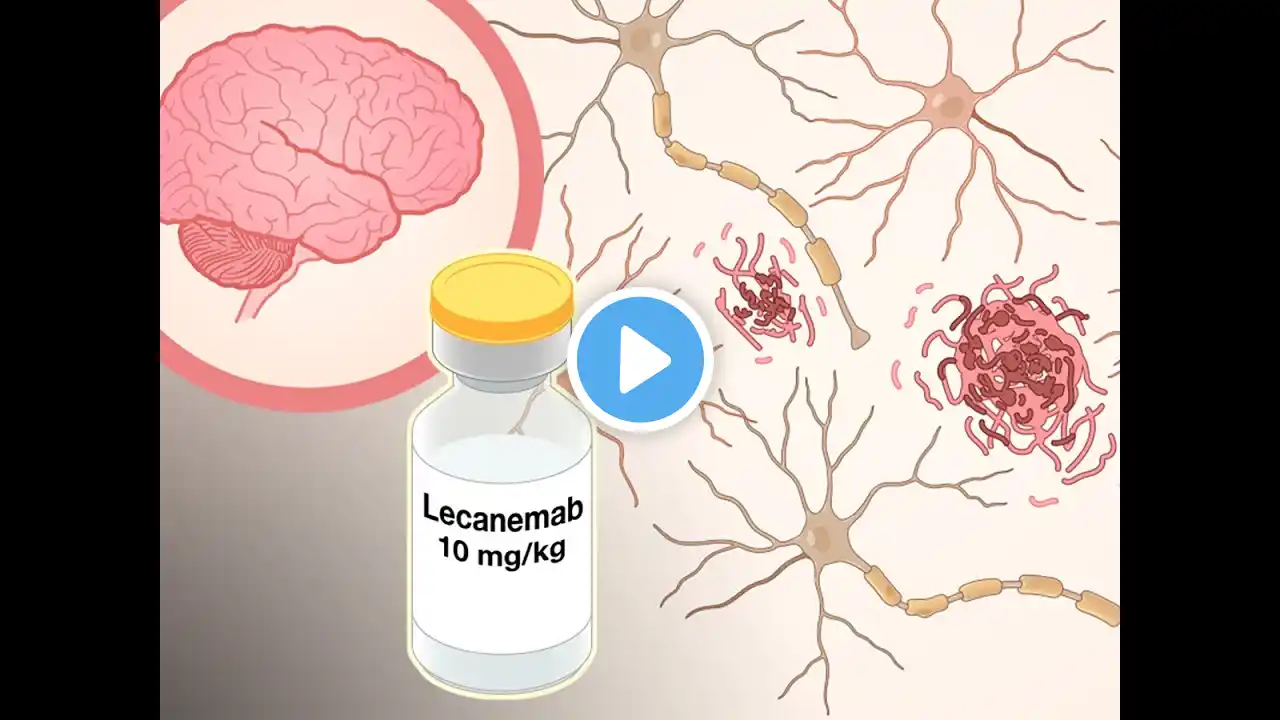
Lecanemab: Breakthrough Alzheimer drug? #alzheimer #alzheimers #alzheimersdisease Japanese drug maker Eisai comes up with new drug Lecanemab for treating Alzheimer's disease. The study is the latest test to a three decade-old theory that Alzheimer's disease is triggered by amyloid beta plaques in patients' brains. And that it can be slowed by drugs that target the build-up of those plaques. Drug companies have struggled through a long list of failed clinical trials with amyloid-targeting drugs. The failure of drug ‘Aduhelm’ last year, and the disappointment it brought to people with Alzheimer's. The positive data also offer Biogen a second chance after the company’s disastrous rollout of another Alzheimer’s drug, ‘Aduhelm’. That medication won regulatory approval last year despite little evidence that it could slow cognitive decline. It received only sharply limited coverage by Medicare and has proved to be a commercial failure. Lecanemab, is administered via intravenous infusion. The trial was the largest to date to test whether clearing the brain of plaques formed by the accumulation of a protein called amyloid could slow the progression of Alzheimer’s disease. Aduhelm is designed to work in a similar way. As with previous anti-amyloid drugs, some patients taking lecanemab experienced brain swelling or brain bleeding. But the prevalence of these side effects was lower than with Aduhelm and other experimental medications. Eisai had already applied for accelerated approval, the type of approval given to Aduhelm. The process allows the Food and Drug Administration to green light drugs if they have uncertain evidence of benefit but affect a disease’s biological pathway in a way that is considered reasonably likely to benefit patients. The company said on Tuesday night that it would first continue with the accelerated approval process, with an F.D.A. decision expected by early January. It will then use the newer data to seek full approval.