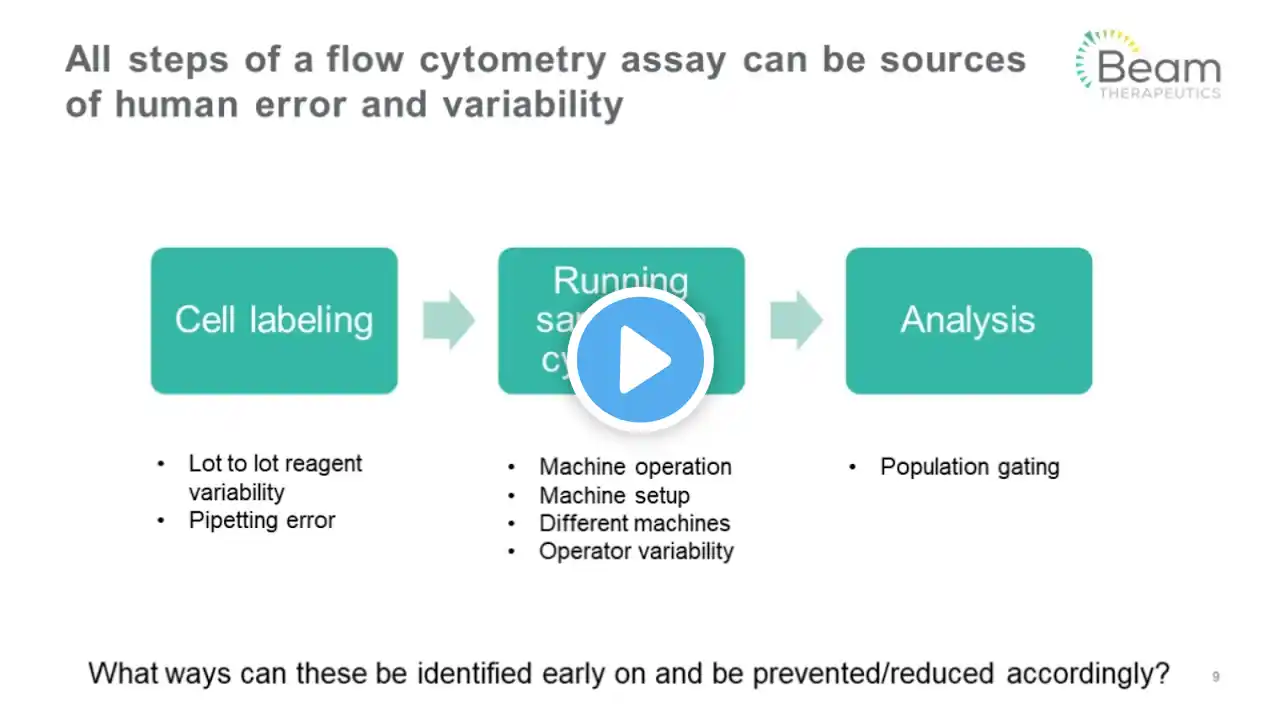
Developing flow-based analytical methods for gene-edited CAR-T programs: How to ensure an...
Presented By: Aaron Edwards Speaker Biography: Aaron received his Masters in Biomedical Sciences from Boston University in 2014, where he worked at the Ragon Institute of MGH, MIT, and Harvard to help develop mRNA vaccines to allow for the production of functionally enhanced antibodies targeting various pathogens. After graduating, he continued his work with this technology at Novartis Vaccines and Diagnostics, exploring ways to improve mRNA vaccines currently under development. His interest in mRNA technologies eventually led him to Bluebird Bio, where he applied his previous learnings on the technology to both gene-edited CAR-T and tgTCR programs. He is now at Beam Therapeutics, where he is the in-vitro biology lead for their first base-edited CAR-T program. Webinar: Developing flow-based analytical methods for gene-edited CAR-T programs: How to ensure an efficient transition from early research to clinical development Webinar Abstract: As cell therapies become more complex, the need for robust analytical tools to characterize such products as they enter the clinic has increased in kind, especially those related to flow cytometry. CAR-T therapies are no exception to this trend, with the next generation of products containing multiple binders, synthetic receptor/ligand combinations, and/or numerous genetic edits. These additional product accoutrement introduce challenges to the already required FACS-based purity, identity, and safety assays needed to begin clinical trials. To ensure a streamlined transition from early research all the way through clinical development, the latter of which is oftentimes performed at external CDMOs, it is critical to establish analytical assays and streamlined processes utilizing machines that can ensure consistent and reproducible data not only across groups within a company but also between production sites. By creating this infrastructure early in the development of next generation cell therapeutics, scientific organization can avoid costly delays and misinterpretation of data that will ensure the most opportunities for the field to successfully take their products to the clinic, hopefully benefitting patients in need. Earn PACE Credits: 1. Make sure you’re a registered member of LabRoots (https://www.labroots.com/webinar/deve...) 2. Watch the webinar on YouTube or on the LabRoots Website (https://www.labroots.com/webinar/deve...) 3. Click Here to get your PACE credits (Expiration date – NOV 04, 2022): (https://www.labroots.com/credit/pace-...) LabRoots on Social: Facebook: / labrootsinc Twitter: / labroots LinkedIn: / labroots Instagram: / labrootsinc Pinterest: / labroots SnapChat: labroots_inc


















