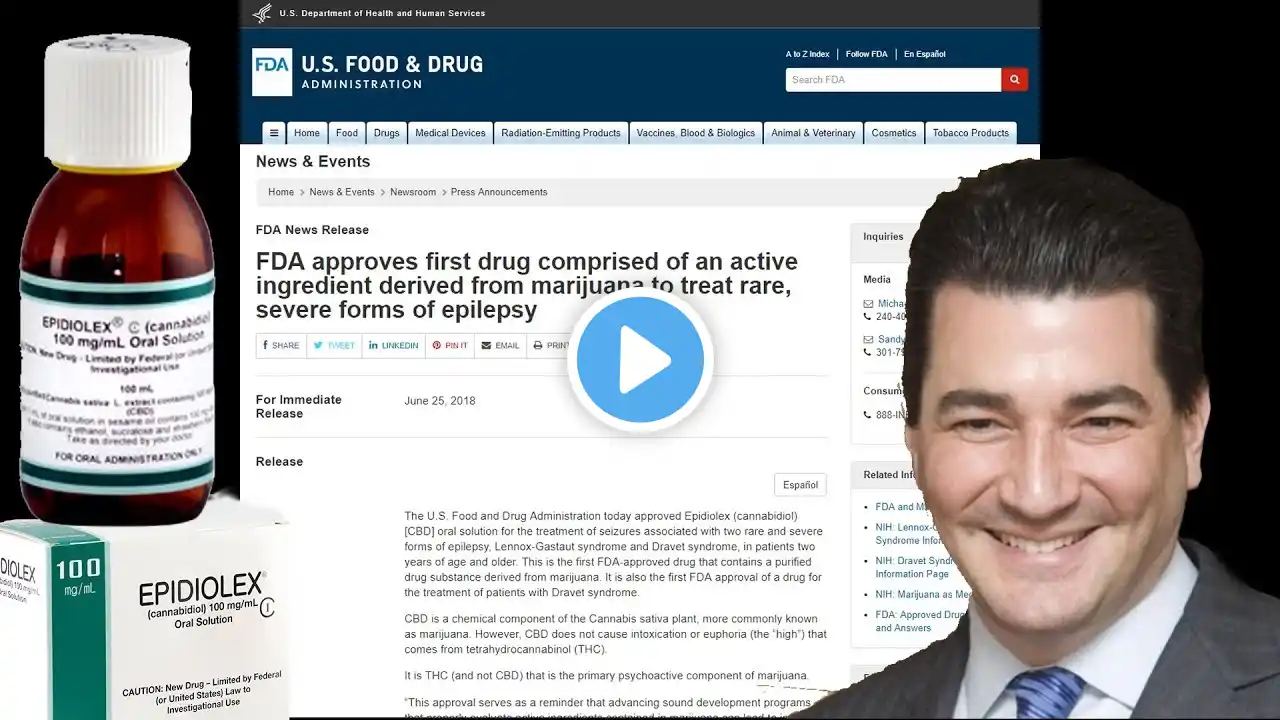
FDA Approves CBD Drug for their Buddies, Threatens a Crackdown for the Rest of Us
In the last stages of the Drug War, we see components individually derived from the Cannabis Plant turned into medicine for specific conditions so that specific Pharma Corporations can make BILLIONS of dollars off of Sick Kids. Welcome to America, 2018. The FDA is Approving Epidiolex, a Pure CBD medicine for 2 kinds of rare epilepsy (Lennox-Gastaut syndrome and Dravet syndrome). GW Pharmaceuticals, plc, also manufactures Sativex, a THC/CBD mouth spray. In a statement about the approval, FDA Director Scott Gottlieb basically sent a threat out to anyone who manufactures a CBD product that isn't Epidiolex. Updates will be sure to come on this as it develops.