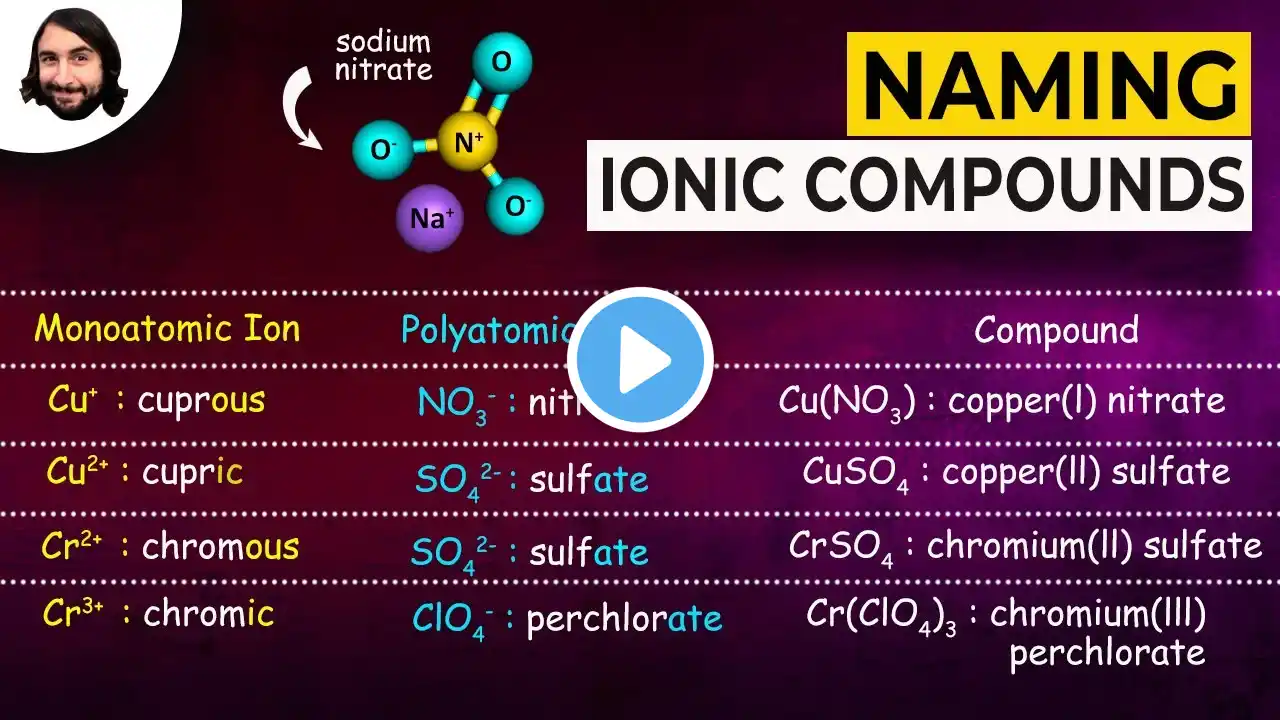
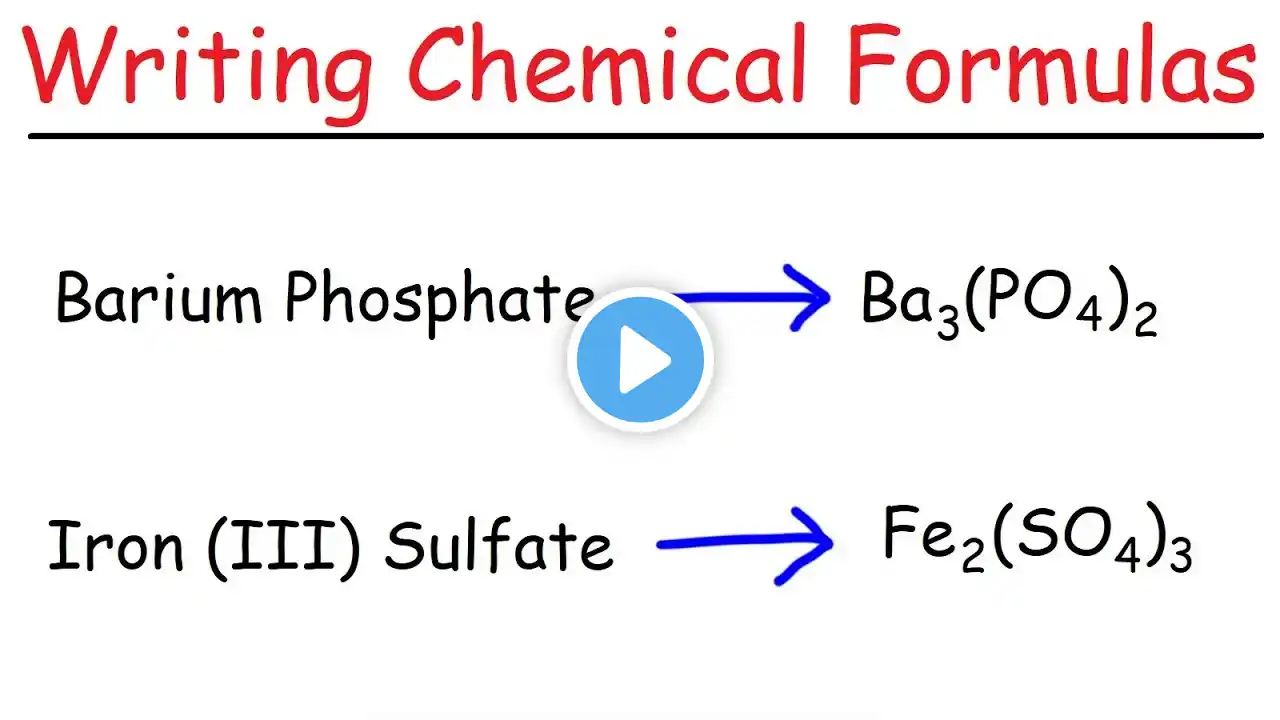
How to Name Ionic Compounds with Transition Metals
In this video we'll write the correct name for ionic compounds with transition metals ions (elements that can have varying ionic charge). Lots more practice at: https://www.breslyn.org/chemistry/nam... To write the name for ionic compounds with transition metals we’ll use the Periodic Table and the Common Ion Table and follow some simple rules. -- Keys for Naming Compounds with Transition Metals-- • Write the name of transition metal as it appears on the Periodic Table. • Write the name and charge for the non-metal. If you have a polyatomic ion, use the Common Ion Table to find and write the formula and charge. • Use the total charge on the non-metal (or polyatomic ion) find the charge on the transition metal. • After the name for the metal, write its charge as a Roman Numeral in parentheses. Example: Iron (III) chloride For a complete tutorial on naming and formula writing for compounds visit: http://www.breslyn.org/chemistry/naming Drawing/writing done in InkScape. Screen capture done with Camtasia Studio 4.0. Done on a Dell Dimension laptop computer with a Wacom digital tablet (Bamboo).