General Chemistry II CHEM-1412 Ch 20 Electrochemistry Part 3 Voltaic Cells
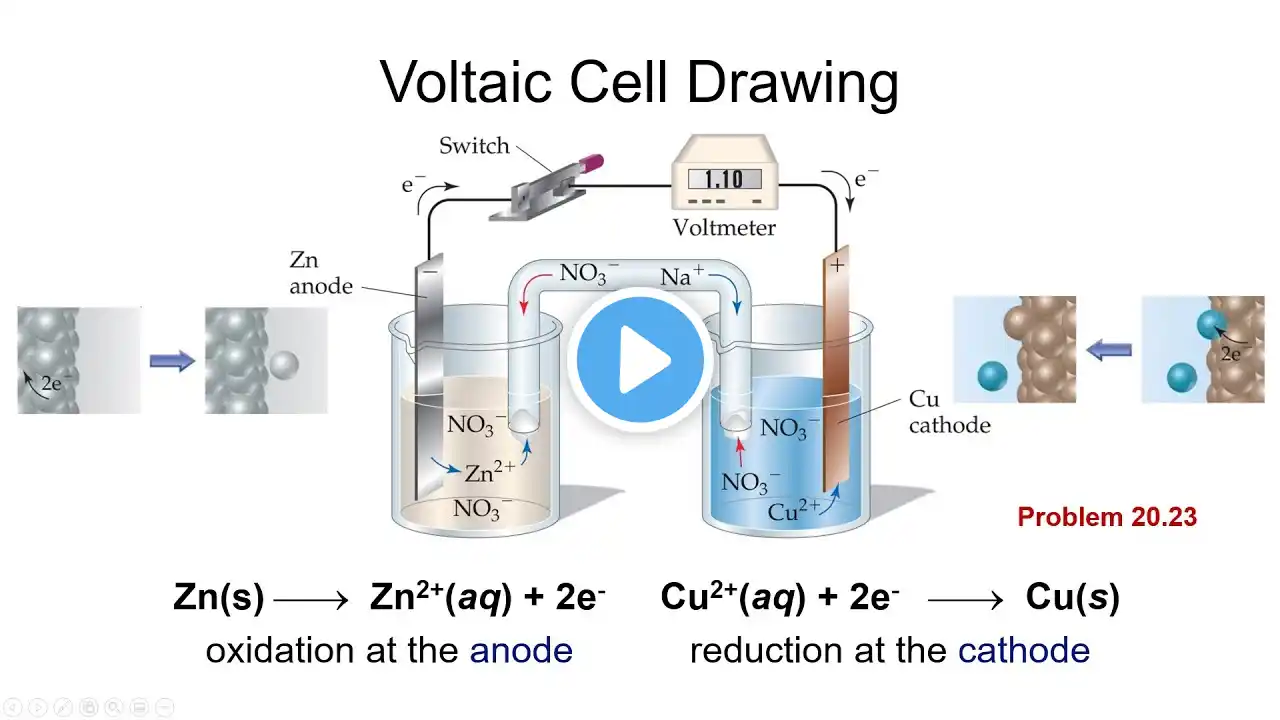
0:00 Section 20.3 Voltaic Cells 4:11 Voltaic Cells Can Do Work 6:53 Voltaic Cell Drawing (study this diagram) 12:24 Example problem: Describe the voltaic cell, identify the direction of electron and ion flow, the signs of the anode and cathode, and the reactions that occur at the anode and cathode. 18:45 Section 20.4 Cell Potentials under standard conditions 20:24 Example problem: What is the meaning of the term cell potential? What is the definition of the volt? 20:51 Electromotive Force (EMF) 22:42 Standard Reduction Potentials 25:52 Standard Hydrogen Electrode 27:33 Calculating Standard Cell Potential 32:46 Example problem: For the oxidation of aluminum metal by chromium(III) ion to form aluminum ion and chromium(II) ion, write the half reactions at each electrode. Calculate the standard cell potential. Sketch the voltaic cell. 42:20 Example problems: Calculate the standard EMF for the given chemical equations. 51:27 For Spontaneous Reactions, Ecell standard greater than 0 V 53:02 Example problem: For a given system, determine which electrode is the anode and which is the cathode. Write the balanced equation and calculate the standard cell potential. 58:54 Stronger Oxidizing and Reducing Agents 1:01:34 Example problems: Based on standard reduction potentials, give the stronger oxidizing agents. 1:04:30 Example problems: Based on standard reduction potentials, give the stronger reducing agents These are the lectures that I use in my General Chemistry II course. The textbook I use is Brown, Theodore et al "Chemistry: The Central Science", Pearson, 14th Ed., 2018, ISBN13: 978-0134414232. Some images used in this video come from that textbook.


















