Kevin Ahern's Bite-Sized Biochemistry #13: Catalytic Strategies II
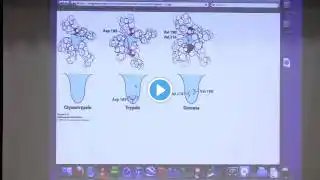
1. Contact - [email protected] 2. Kevin's lectures with The Great Courses - https://www.thegreatcoursesplus.com/b... 3. Kevin's Lecturio videos for medical students - https://www.lecturio.com/medical-cour... 4. Course materials at https://kevingahern.com/biochemistry-... 5. Course video channel at • Kevin Ahern's Bite-Sized Biochemistry #1: ... 6. Metabolic Melodies at https://teeheetime.com/category/lyric... 7. Kevin's Free Biochemistry books - https://kevingahern.com/biochemistry-... 8. Kevin's Pre-med Audio course on Listenable - https://listenable.io/web/courses/143... Lecture Highlights Catalytic Mechanisms II 1. The S1 pocket determines a serine protease's specificity. The S1 pocket of chymotrypsin is hydrophobic and relatively large, allowing it to bind phenylalanine, for example. Remember that chymotrypsin cuts adjacent to phenylalanine (among other hydrophobic amino acids). The S1 pocket of trypsin, for example has a negatively charged group in the bottom, allowing it to bind to lysine or arginine. 2. Other proteases include cysteine proteases (use cysteine and histidine in the active site), aspartyl proteases (use aspartic acids and water in the active site) metalloproteases (use a metal ion - usually zinc - and water in the active site). 3. Cysteine proteases use an ion of the sulfhydryl group of cysteine to act as a nucleophile to attached the carbonyl peptide bond and facilitate breakage of the peptide bond. 4. Aspartyl proteases use two aspartic acid side chains to hold water in place and use an ion of it to act as a nucleophile to attack the peptide bond. 5. Metalloproteases use a metal ion to hold water in place so it can be ionized to act as a nucleophile to attack the peptide bond. 6. Carbonic anhydrase is an enzyme that catalyzes the joining of carbon dioxide and water to form carbonic acid. 7. A zinc ion (held in place by three histidines in the active site of carbonic anhydrase) plays an important role in the catalysis of the enzyme by binding a water molecule. A subsequent loss of a proton by water is necessary for catalysis. Notably, the enzyme has maximal activity at a high pH (where protons are easily removed) and a lower activity in an acidic pH (6.0). 8. The limiting step in the action of carbonic anhydrase is the abstraction of the proton from water. Buffers and/or bases help facilitate this and thus speed the reaction. 9. Restriction enzymes are bacterial enzymes that can cleave DNA by breaking phosphodiester bonds between adjacent nucleotides in the molecule.