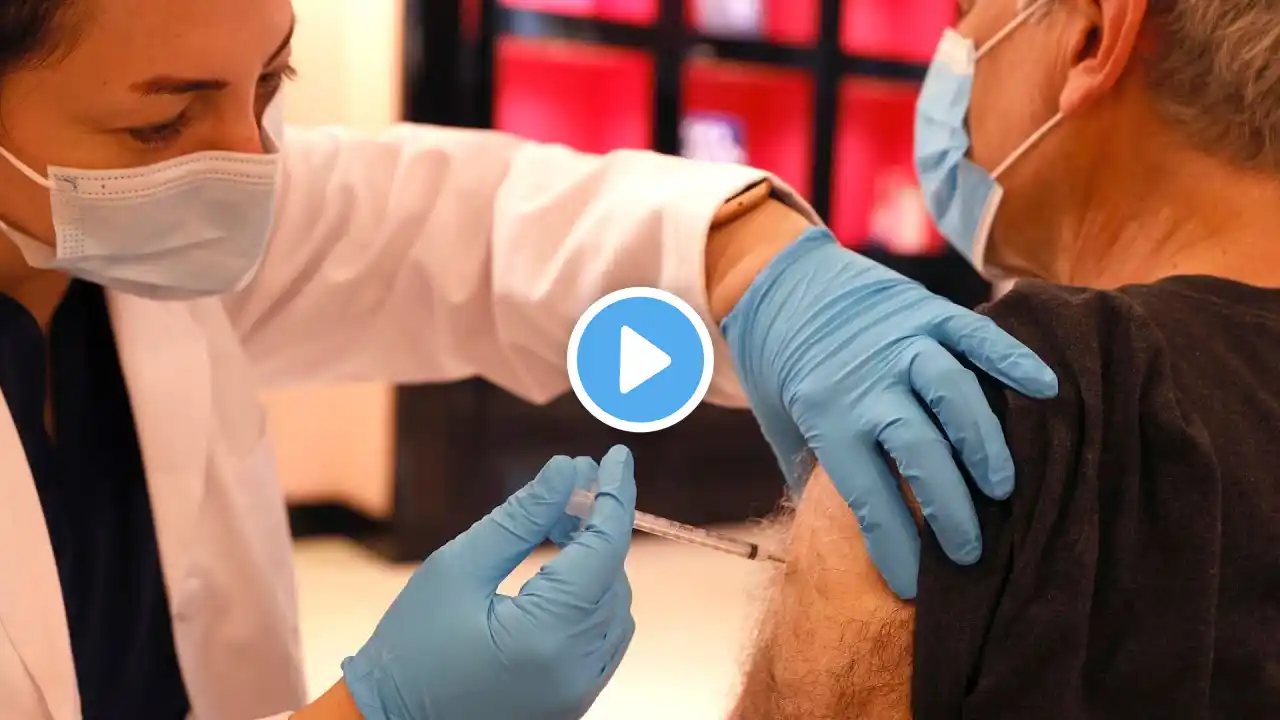
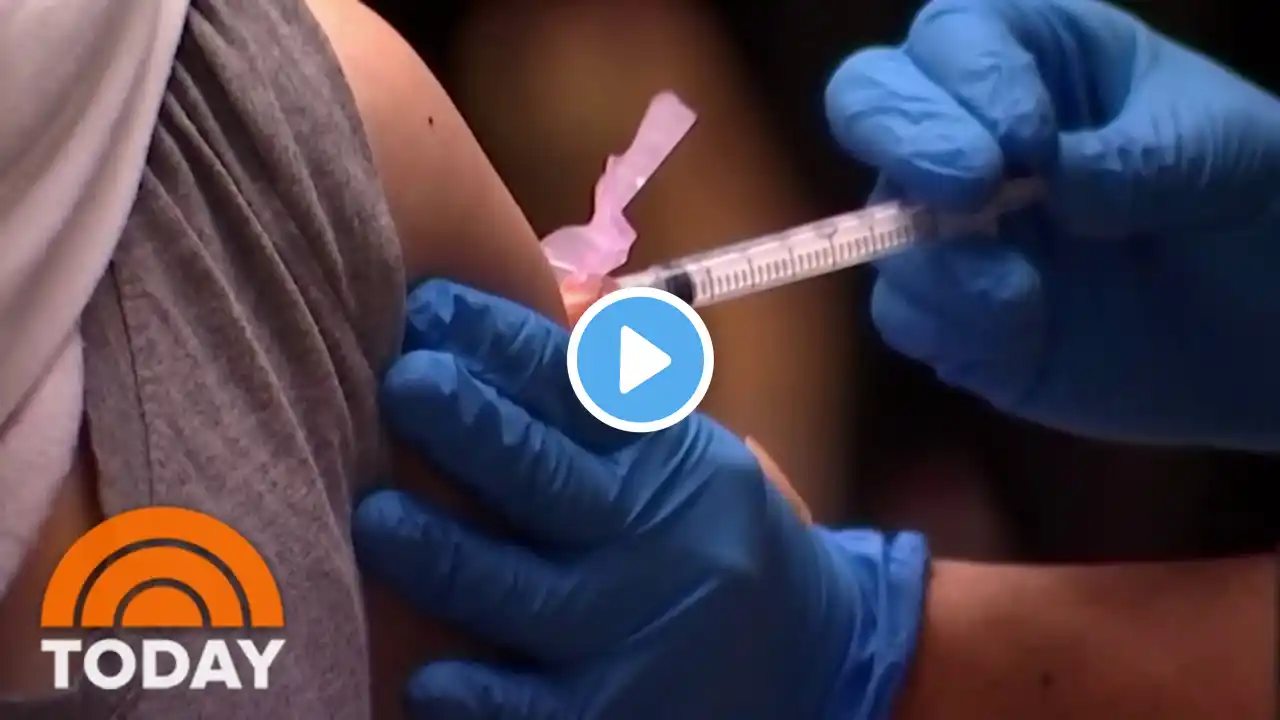
FDA Clears Pfizer Booster Shots for 12 to 15-Year-Olds
The U.S. Food and Drug Administration (FDA) announced its authorization for use of a booster dose on Jan. 3. Fully vaccinated children in the age range will become eligible for a booster shot in May. COVID-19 booster shots may help provide better protection against both the delta and omicron variants. In authorizing a third shot for this age range, health experts with the FDA cited the apparent increased resistance of the omicron variant to two vaccine doses. In addition, the FDA shortened the time span between second and third shots by a month. To watch BNC programming, visit https://bnc.tv/how-to-watch/ Follow BNC on social media: Facebook: / bncnews Twitter: / bncnews Instagram: / bncnews #FDA #Boostershots #Pfizer