COVID-19 ECHO Session #33: Testing Strategy for COVID-19 Antigen Rapid Diagnostic Tests
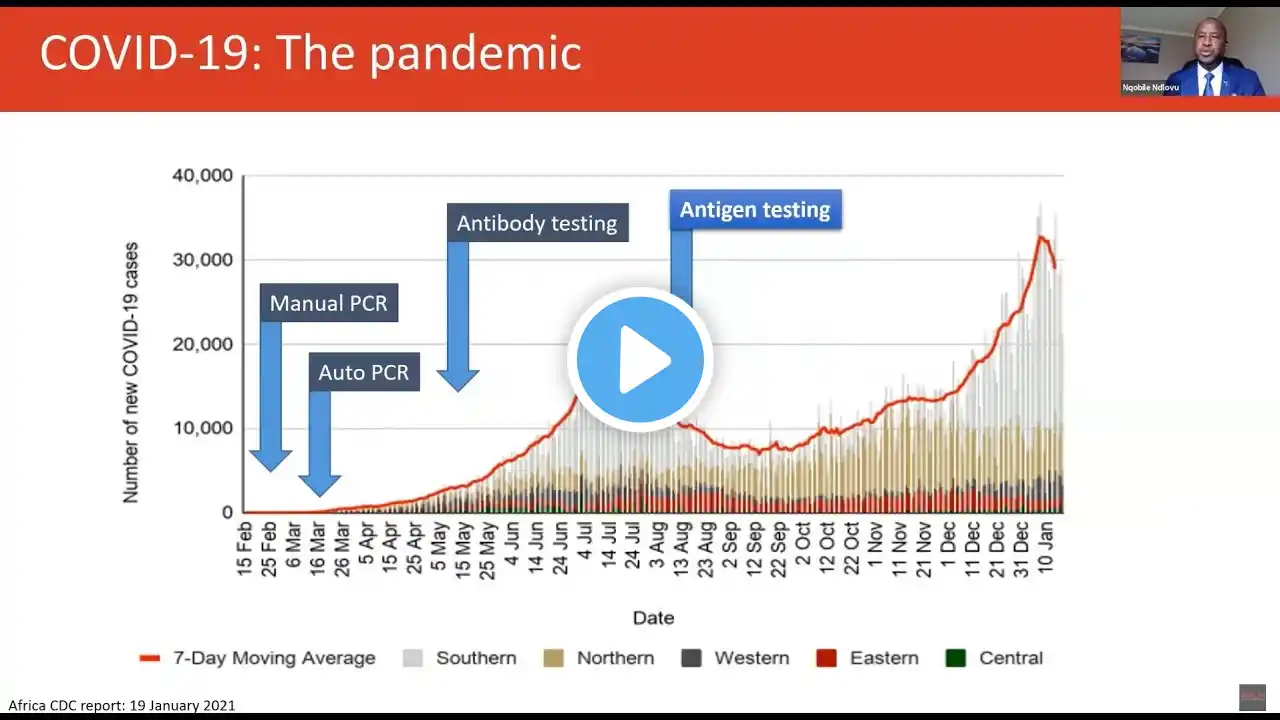
This session focuses on the details of Africa CDC’s antigen rapid diagnostic testing guidelines and recommendations. The start of 2021 has seen new challenges arise in the COVID-19 pandemic, with rapidly growing spread and new virus variants. To urgently address this, many countries are now deploying rapid antigen tests and shifting these assays into more widespread use. In an effort to support this strategy, the Africa CDC has recently released guidelines to expand the use of COVID-19 rapid antigen tests. The guidelines are available on Africa CDC website mentioned below and the announcement is available below as well. The aim of the Africa CDC guidelines is to provide Member States with recommendations on testing approaches and algorithms to help curb the growing spread of SARS-CoV-2. The guidelines are much needed at this critical time to support the design of strategies and implementation plans for testing within the health and non-health sectors, and for testing within communities. In this session we will: • unpack the algorithms in the context of testing in different settings • address questions around how best to deploy antigen tests and related tools for more effective epidemic control Speakers: Nqobile Ndlovu, CEO, ASLM Yenew Kebede, Head, Laboratory Systems & Networks, Africa CDC Sikhulile Moyo, Deputy Laboratory Director, Botswana-Harvard Partnership Websites Guidelines: https://africacdc.org/download/interi... Announcement: https://africacdc.org/news-item/new-g...