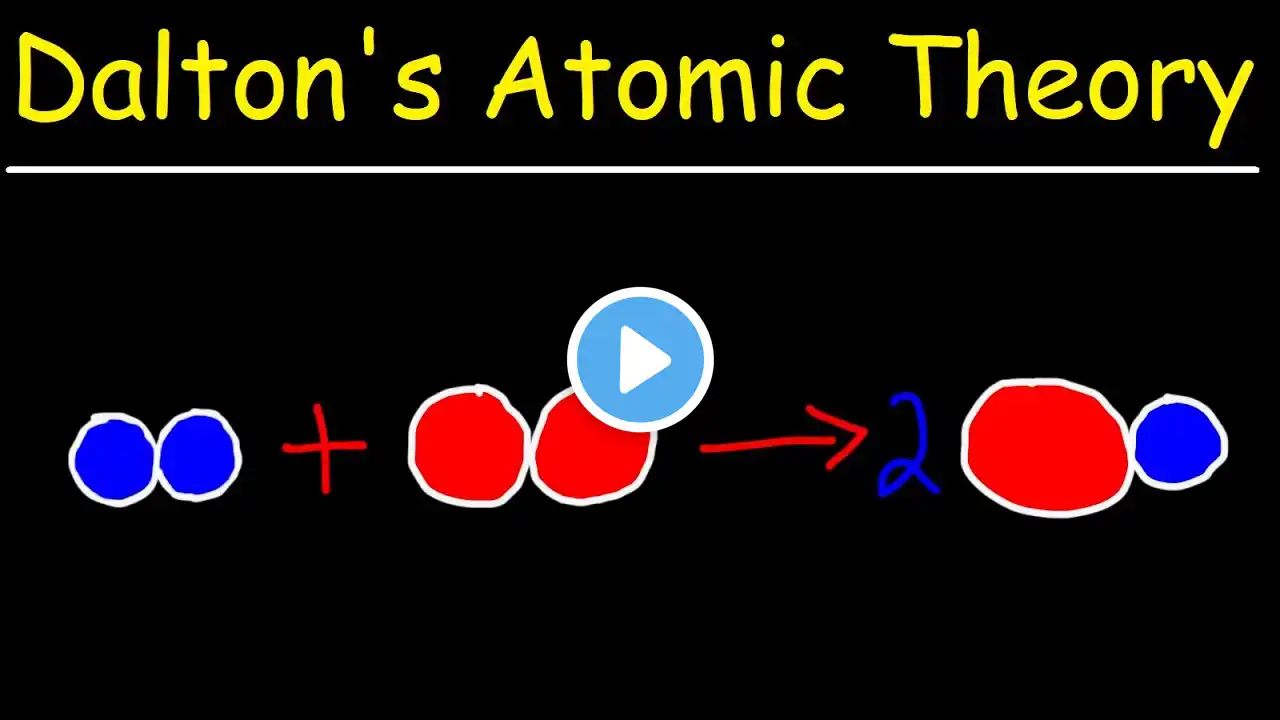
a. In 1803, John Dalton proposed the first atomic theory, in which he stated that atoms could not b…
a. In 1803, John Dalton proposed the first atomic theory, in which he stated that atoms could not be divided into smaller units of matter. Although that part of his theory has been proven wrong, his theory is consistent with the law of conservation of mass (matter). i. What does the law of conservation of matter state? ii. How are changes between phases consistent with the law of conservation of matter? b. Atomic theory has evolved over time as new discoveries have been made about the nature of matter. Use the following questions to highlight some of these discoveries. i. How did J. J. Thomson #x27;s bent cathode-ray experiment change Dalton #x27;s model of the atom? ii. How did Ernest Rutherford #x27;s gold-foil experiment change Thomson #x27;s model of the atom? Watch the full video at: https://www.numerade.com/ask/question... Never get lost on homework again. Numerade is a STEM learning website and app with the world’s largest STEM video library. Join today and access millions of expert-created videos, each one skillfully crafted to teach you how to solve tough problems step-by-step. Join Numerade today at: https://www.numerade.com/signup/