Isoprene
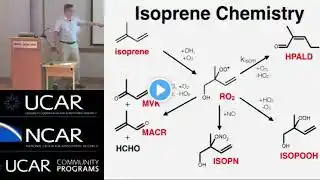
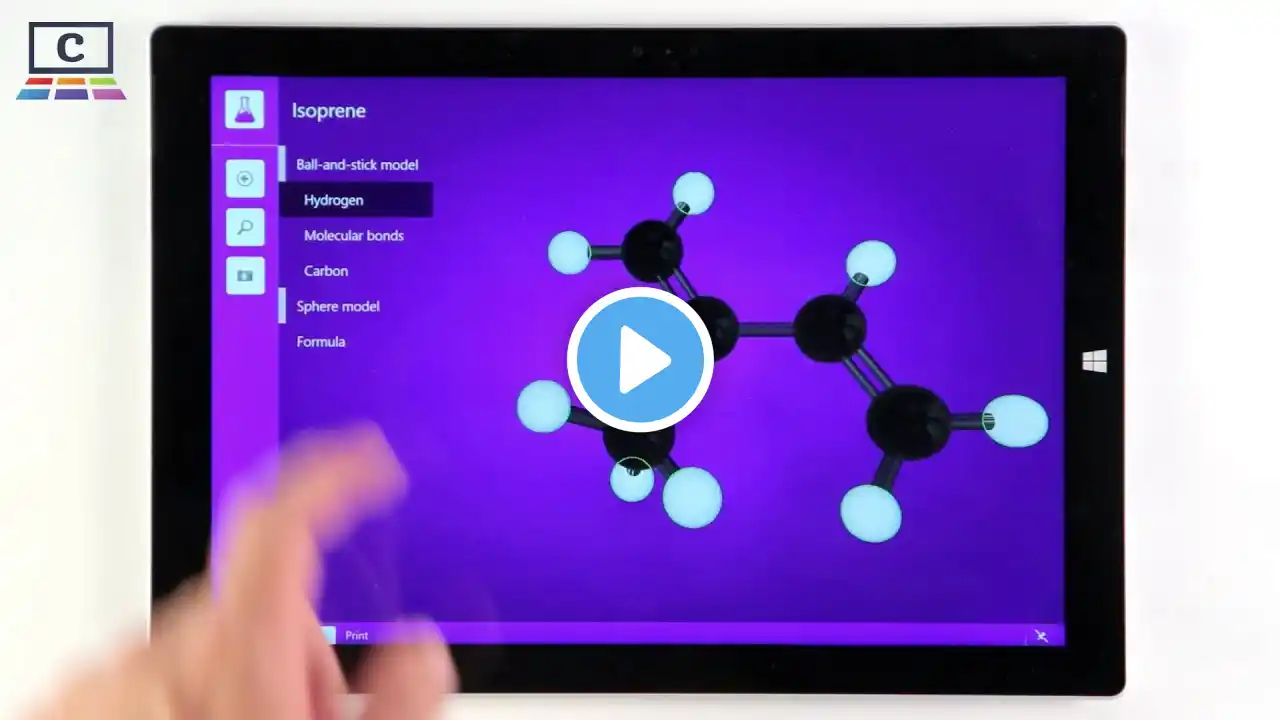
Except where otherwise noted, data are given for materials in their standard state. Isoprene, or 2-methyl-1,3-butadiene, is a common organic compound with the formula CH2=C-CH=CH2. In its pure form, it is a colorless volatile liquid. Isoprene is produced by many plants. Isoprene is produced and emitted by many species of trees. Yearly production of isoprene emissions by vegetation is around 600 million tonnes, half from tropical broadleaf trees and the remainder primarily from shrubs. This is about equivalent to methane emissions and accounts for ~1/3 of all hydrocarbons released into the atmosphere. Isoprene is made through the methyl-erythritol 4-phosphate pathway in the chloroplasts of plants. One of the two end products of MEP pathway, dimethylallyl pyrophosphate, is catalyzed by the enzyme isoprene synthase to form isoprene. Therefore, inhibitors that block the MEP pathway, such as fosmidomycin, also blocks isoprene formation. Isoprene emission increases dramatically with temperature and maximizes at around 40°C. This has led to the hypothesis that isoprene may protect plants against heat stress. Emission of isoprene is also observed in some bacteria and this is thought to come from non-enzymatic degradations from DMAPP. Isoprene emission in plants is controlled both by the availability of substrate and by enzyme activity. In particular, light, CO2 and O2 dependencies of isoprene emission are controlled by substrate availability, whereas temperature dependency of isoprene emission is regulated both by substrate level and enzyme activity. Isoprene is the most abundant hydrocarbon measurable in the breath of humans. The estimated production rate of isoprene in the human body is 0.15 µmol/, equivalent to approximately 17 mg/day for a person weighing 70 kg. Isoprene is common in low concentrations in many foods. Isoprene emission appears to be a mechanism that trees use to combat abiotic stresses. In particular, isoprene has been shown to protect against moderate heat stress. It may also protect plants against large fluctuations in leaf temperature. Isoprene is incorporated into and helps stabilize cell membranes in response to heat stress. Isoprenes are used in the cell membrane monolayer of many Archaea, filling the space between the diglycerol tetraether head groups. This is thought to add structural resistance to harsh environments in which many Archaea are found. The isoprene skeleton can be found in naturally occurring compounds called terpenes, but these compounds do not arise from isoprene itself. Terpenes can be viewed as multiples of isoprene subunits, and this perspective is the cornerstone of the 'isoprene rule'. The precursor to isoprene units in biological systems is dimethylallyl pyrophosphate and its isomer isopentenyl pyrophosphate. The plural 'isoprenes' is sometimes used to refer to terpenes in general. Isoprene chains are commonly found in numerous biologically active oligomers such as Vitamin A. Similarly, natural rubber is composed of linear polyisoprene chains of very high molecular weight and other natural molecules. After release, isoprene is converted by free radicals and to a lesser extent by ozone into various species, such as aldehydes, hydroperoxides, organic nitrates, and epoxides, which mix into water droplets and help create aerosols and haze. Video Empire produces videos read aloud. Use the information in this video at your own risk. We cannot always guarantee accuracy. This video uses material from https://en.wikipedia.org/wiki/Isoprene, licensed with CC Attribution-ShareAlike 3.0. This video is licensed with CC Attribution-Share-Alike 3.0 https://creativecommons.org/licenses/... In order to adapt this content it is required to comply with the license terms. Image licensing information is available via: https://en.wikipedia.org/wiki/Isoprene