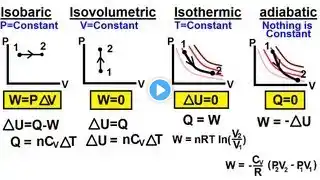
Thermodynamics - Problem 4
One mole of an ideal gas expands at a constant temperature of 300 K from an initial volume of 10 litres to a final volume 20 litres. The work done in expanding the gas is (R=8.31 J/mole K) (a) 750 J (b) 1728 J (c) 1500 J (d) 3456 J